Severe asthma is defined as asthma that remains uncontrolled with on-going symptoms despite maximal therapy and where other reasons have been addressed, such as improving adherence to treatment, addressing technique issues with inhaler use, decreasing triggers and other factors (e.g. smoking, weight management, chronic sinusitis).
It is thought that approximately 3% of asthmatics are defined as having severe asthma.
People with severe asthma usually still have persistent symptoms and frequent episodes of breathlessness, despite being on maximal therapy. Typically, this therapy would be a high dose inhaled corticosteroid and long acting (beta agonist) controller. Common combination medications used include: Seretide, Symbicort, Flutiform, Breo Ellipta, Fostair and DuoResp Sprimax.
Severe asthma can significantly impact quality of life by reducing physical activity, negatively impacting personal and family relationships, as well as those at work. There is also an increased number of other co-existing illnesses in patients with severe asthma that can add further to the risk of hospitalisation and mood disorders (such as depression or anxiety).
If common factors that contribute to poor asthma control have been addressed (as outlined above) and maximal inhaled therapies are being used, but you your asthma is still uncontrolled, you may be eligible for monoclonal antibody therapy.
Monoclonal antibody therapy is used as an add-on treatment to your usual therapy (meaning you continue to use your current inhaled medications whilst on it). It can be particularly helpful in those with severe (eosinophilic) asthma with an allergic component. Persistent eosinophil inflammation is seen in more than half of all patients with severe asthma. Several monoclonal antibodies targeting eosinophil inflammation have been developed to treat people with asthma of an eosinophilic type, including 2 targeting either interleukin-5 (IL-5) or its receptor (IL-5R), benralizumab and mepolizumab.
However, there are currently 4 monoclonal antibody therapies available in Australia:
- Benralizumab (Fasenra)
- Mepolizumab (Nucala)
- Dupilumab (Dupixent)
- Omalizumab (Xolair)
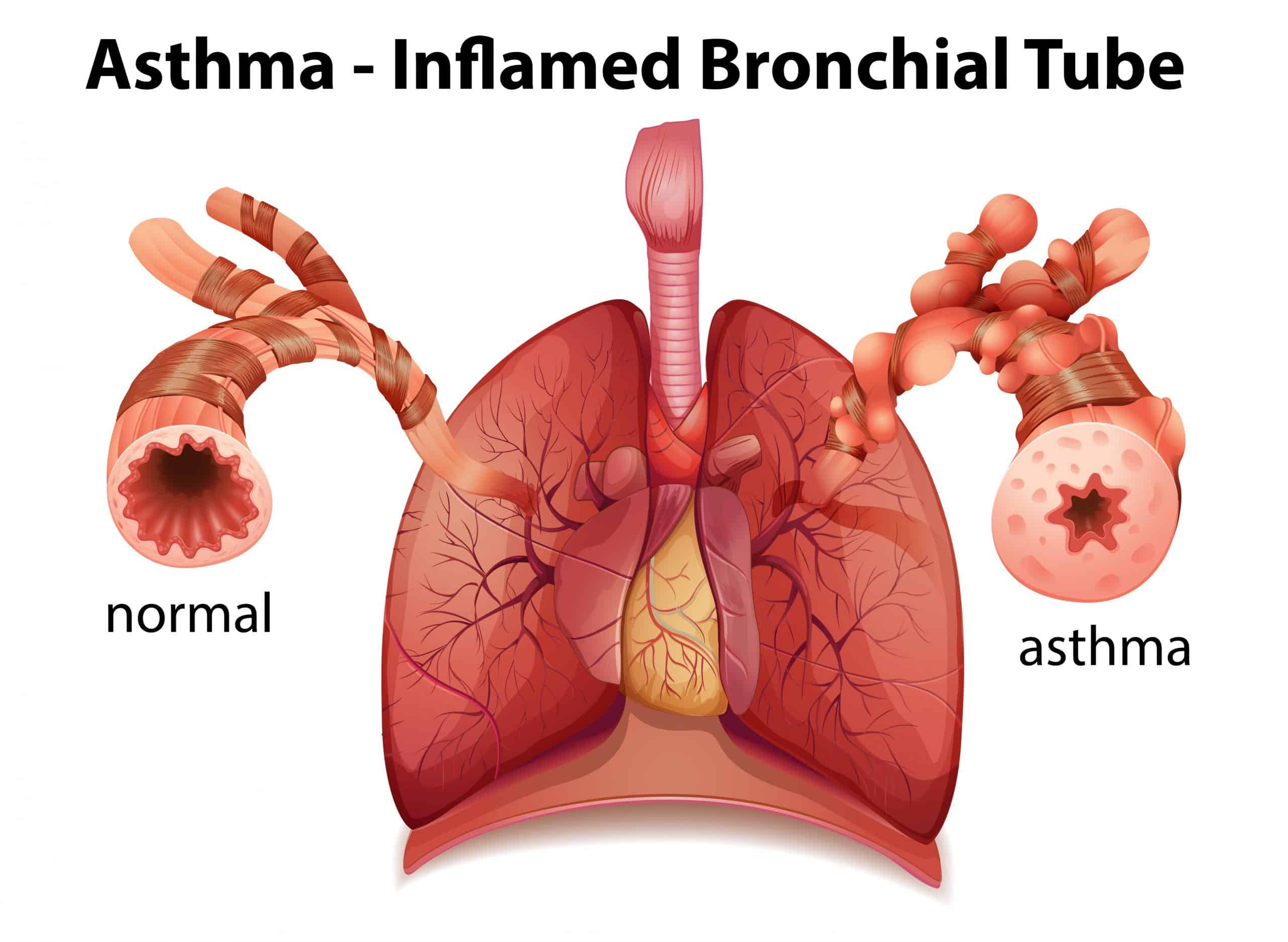
These medications work by reducing the number of white blood cells called eosinophils in your blood and lungs. This type of white blood cell is associated with inflammation of the airways, resulting in worsening of asthma symptoms. Patients with asthma generally have higher than normal levels of eosinophils than their non-asthmatic counterparts.
These medications are available on the Pharmaceutical Benefits Scheme (PBS) for adults and adolescents over the age of 12. They are given as subcutaneous injections (self-injected under the skin) at varying time intervals ranging from once every 2 weeks to once every 4-8 weeks. At present you would need a referral from your GP to a specialist (i.e. respiratory physician or clinical immunologist) and be under their care for 6 months before becoming eligible for the PBS subsidy.
It is important to still check in with your local doctor and make sure you have a current Asthma Management Plan in place and that it is updated annually.
More information on severe asthma and its management can be found on the Asthma Australia website:
https://asthma.org.au/about-asthma/severe-asthma/
Advice on and videos showing good inhaler technique is available on the National Asthma Council website:
In addition, the National Asthma Council also has more on the use of monoclonal antibody therapy for severe asthma:
Here at AusTrials, we will soon be studying a potential new add-on treatment for severe asthma. Contact AusTrials on 1300 190 841 for more information.

